Category Archives: DMH News
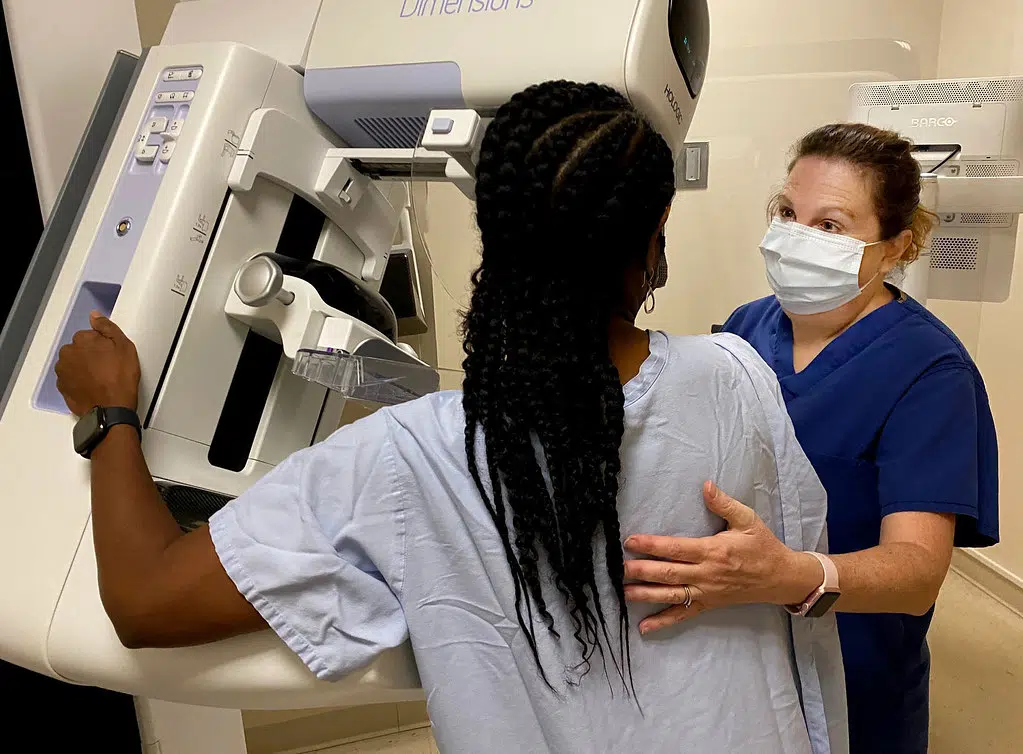
Memorial to offer free mammograms in October
October 4, 2023 - Free breast cancer screenings will be available at all five Memorial Health hospitals for women aged 40 and older who are uninsured or underinsured during October as part of Mammogra...
Lindsay Romano Oct 04, 2023 Navy Medicine

LISTEN: DMH discusses healthcare industry changes on Byers & Co
October 4, 2023 - Drew Early and Dr. Ted Clark of DMH joined Byers & Co to talk about industry challenges, changes since COVID-19, and their investment and focus on serving the community. Listen t...
Lindsay Romano Oct 04, 2023

DMH to host free Community Education Event for Breast Cancer Awareness Month
September 26, 2023- Decatur Memorial Hospital will be hosting its next installment of the Memorial Specialty Care Community Education Series. General surgeon Dr. George Liu will present "Breast Cancer...
Dominique Bates-Smith Sep 26, 2023

Free Memorial Health prostate cancer screening set for Sept. 22
September 18, 2023 - Memorial Health and Cancer Care Specialists of Illinois are co-sponsoring free prostate cancer screenings this coming Friday, Sept. 22, through SIU Urology at Decatur Memorial Hos...
Lindsay Romano Sep 18, 2023

Foley named Memorial Health Ambulatory Colleague of the Month for August
September 18, 2023 -Cartha Foley, RN, Memorial Specialty Care Gastroenterology at Decatur Memorial Hospital, has been named the Memorial Health Ambulatory Group Colleague of the Month for August. Fole...
Lindsay Romano Sep 18, 2023

Sarah German with CureACC Donates Cancer Care Packages to Decatur Memorial Hospital
September 11, 2023 -Sarah German, 18, is a young woman with a heartfelt mission. Her mom's fight against adenoid cystic carcinoma (ACC) prompted her to start CureACC, a nonprofit organization that str...
Lindsay Romano Sep 11, 2023

LISTEN: DMH Foundation's Julie Bilbrey on Byers & Co
September 6, 2023 - Julie Bilbrey of Decatur Memorial Foundation joined Byers & Co to talk about funds raised through Par-For-The-Cure, and KerbyStrong 100 Race Day. Listen to the podcast now!
Lindsay Romano Sep 06, 2023

General Surgeon Dr. Mohammad Torabi Joins Memorial Specialty Care
August 28, 2023 - General surgeon Dr. Mohammad Torabi has joined Memorial Specialty Care in Decatur. Dr. Torabi earned his medical degree from American University of Antigua before serving a residency...
Lindsay Romano Aug 28, 2023

Memorial Health welcomes 3 new critical care providers to Decatur
August 21, 2023 - Three new critical care physicians have joined Decatur Memorial Hospital's Specialty Care team. Critical care physicians have expertise in caring for patients with life-threatening i...
Brandon Kondritz Aug 21, 2023

Frank Hardy named Decatur Memorial Hospital's July Colleague of the Month
August 3, 2023 - Decatur Memorial Hospital has named its July Colleague of the Month, a shuttle driver with the hospital's Patient Experience division. Frank Hardy's colleagues said he's thorough abou...
Brandon Kondritz Aug 03, 2023